Often, we hear about osmosis in the context of some new technology, including artificial intelligence platforms1 (AI). However, every organic system has been using osmosis since the dawn of time. Clearly, osmosis is an important process across all sciences and, in particular, in biology. This article describes osmosis and how it works, and gives you examples you can relate to and experiments to try.
What Is Osmosis?
- The term describes the net movement of water molecules across a semipermeable membrane.
- It works to balance solute concentrations on both sides of the membrane.
- It differs from diffusion in several important ways, even though it is considered a type of diffusion.
- Several factors affect the osmotic process, including pressure and concentration.

What Is Osmosis?
Osmosis is the act of equalising solute concentrations on both sides of a semipermeable barrier.
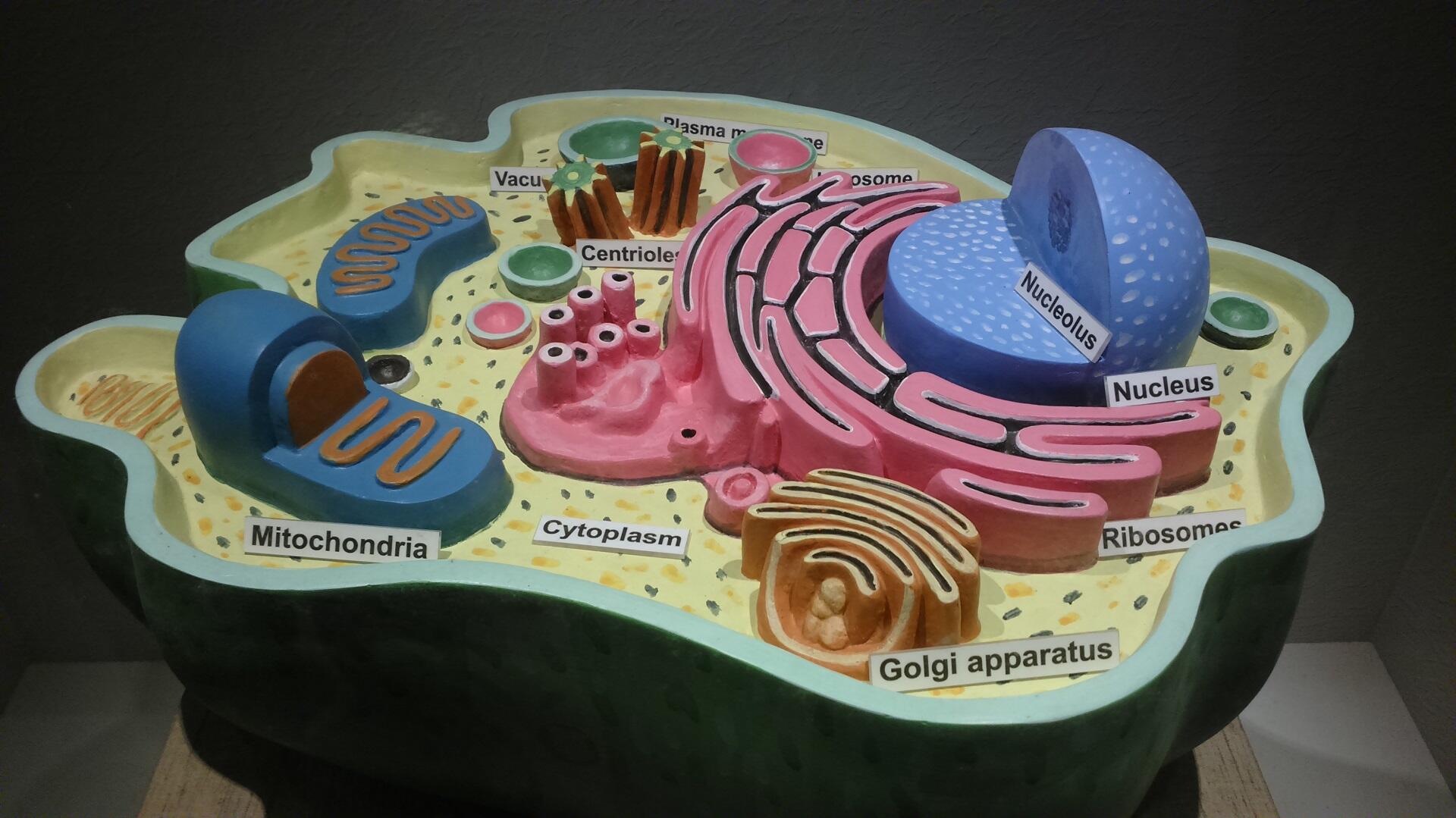
This image shows the structure of an animal cell. Its outermost layer is the cell membrane (also called the plasma membrane), the semipermeable barrier in question.
Should the interior of the cell have a high concentration of solutes, there must be less water present within.
By default, there must also be a lower concentration of solutes - thus, a higher potential for water, outside the cell membrane. That being the case, water molecules will cross the cell wall to adjust the concentration of solutes inside and outside the cell.
This overview helps us envision the osmotic process. Osmosis works the same way in biology as it does in chemical, agricultural, and industrial applications.
Many water filters use a type of osmosis called reverse osmosis. The semi-permeable barrier retains solids larger than water molecules, allowing only H2O to pass through.
Remember that osmosis is a widely used process in manufacturing and other aspects of our daily lives; it doesn't apply only to biology. Often these osmotic processes use solvents and solutes - hence, the use of those words in the general description above.
In fact, the osmotic phenomenon has an entire glossary of words to describe its functions. This chart explains them all.
| 📖Term | 📃Definition | 🔎Example |
|---|---|---|
| hypotonic | the cell gains water molecules. | Soaking raisins in water. |
| turgid | the cell's state when hypotonic. | Raisins, after being soaked in water. |
| isotonic | no net movement of water across the membrane. | Plasma, tears, sweat, etc. |
| flaccid | the cell's state when isotonic. | Plants, when their soil has too high a saline content. |
| hypertonic | the cell loses water molecules. | Using a food dehydrator. |
| plasmolyzed | the cell's state when hypertonic. | Shrunken fruit after dehydration. |
| turgor pressure | the force that presses the plasma membrane within the cell toward the cell wall. | Fresh veg has strong turgor pressure. As they wilt, they lose the pressure. |
| osmotic gradient | the difference between two solutions' concentration across the membrane. | Kidney function to retain fluids and maintain electrolyte balance. |
| osmotic pressure | the minimum pressure needed to prevent inward flow across the semi-permeable membrane. | Water filters (reverse osmosis) |
The Difference Between Diffusion and Osmosis
Thumbing through your biology textbook, you might have noticed that 'diffusion' and 'osmosis' see apparently indiscriminate usage. If that's left you scratching your head, you're not alone: many don't quite get the difference between diffusion and osmosis. This table lays out the differences between these phenomena.
| 💦Osmosis | ⛲Diffusion |
|---|---|
| osmosis fulfils a primary function. | diffusion fulfils a primary function. |
| not associated with mineral or nutrient uptake. | assists cells' mineral and nutrient uptake. |
| only happens in liquids | happens in all states of matter |
| water is required. | no water required. |
| needs a semipermeable membrane. | no semipermeable membranes needed. |
| only solvent molecules move across barriers. | all the particles move across barriers. |
| particle flow in one direction. | particle flow in all directions. |
| depends on the number of solute particles. | depends on having other particles present. |
| the process can be stopped. | the process cannot be stopped. |
In principle, diffusion and osmosis represent the same thing: equalising the concentration of solutes. However, while diffusion may happen in all conditions, osmosis takes place only when there is a semipermeable barrier to cross.
Besides the presence of such a membrane, the major difference between diffusion and osmosis is that, in the latter, only water crosses the membrane. In diffusion, both the solute and solvent particles move to equalise the concentration gradient.
In other fields and sciences, osmosis may take place using other solutes. However, in biology, the solute in question is always water.
In all cases, osmosis is a special type of diffusion, one that requires the presence of a semipermeable membrane that only the solute may cross. Elsewhere, diffusion's purpose in organisms is much broader. Take the time to discover how it works in plants and animals, and which factors affect diffusion.
Osmosis in Biological Systems
I learned from admiration and osmosis.
Joni Mitchell, American songstress.
Have you ever gone on holiday and, upon your return, found your plants all wilted? But then, after giving them water, they perked up again... that is osmotic pressure at work. Plants use osmotic pressure to keep their shape.
When you become dehydrated, the cells in your body undergo the same process. Water molecules from muscles and other organs transfer into your blood cells via osmosis because they are hypertonic. That leaves a high concentration of solutes in those cells, a condition that could disturb their function.
Just like overwatering plants, we cannot simply pour buckets of water down our throats and hope for instant osmotic results. The effects could be downright dangerous2. Slow rehydration is needed to give our cells time to adapt to osmotic pressure.

Factors that Affect Osmosis
Two main factors affect the osmotic process: the concentration gradient (also called the osmotic gradient) and osmotic pressure. You might have gotten the idea from the explanation of hypertonic and hypotonic states above. Here, we dive deeper into these concepts.
Concentration Gradient
The concentration gradient relates to the concentration of particles dissolved in a solution; particularly, the difference between the percentages of concentration.
This condition exists when there is a heavier concentration of solutes on one side of a semipermeable membrane than on the other.
Imagine you've just added a bit of salt to a glass of water. The point where you introduced the salt has a higher concentration gradient than elsewhere in the water.
As the salt sinks to the bottom and accumulates, that's where the concentration is highest. As you then stir the salt into the water, those molecules disperse, ultimately distributing the salt evenly throughout the water.

At that point, the solution is said to have reached equilibrium.
Cells use concentration gradients to move a substance in or out of their cells.
The stored energy inherent in concentration gradients is used for that purpose.
Breathing presents the most obvious example of concentration gradient usage, despite being a diffusion process. Granted, it's an instance of gas reflecting a concentration gradient, not water. Still, it serves well to illustrate the principle.
The oxygen is again diffused from a high-concentration area (the cells) to low-concentration areas - our organs and tissues. The WACE biology tutors near me are brilliant at explaining this phenomenon.
Osmotic Pressure
Osmotic pressure describes how hard water has to push to get through the semipermeable barrier. The concentration of solutes determines osmotic pressure, as water will work harder to access an area with a higher solute concentration.
Of course, water has no conscious knowledge of solute gradients. It obeys the laws of nature that dictate the random distribution of matter. We humans, understand this pressure as a mathematical equation3:
That is, indeed, the symbol for Pi but, in this equation, it does not represent that value. It was adopted by concurrence; chemists all agreed that it served well to represent osmotic pressure. In this equation, solving for π is the same as solving for X in any other algebraic equation.
On the other side of the equal sign, we find:
- i: the number of solute particles in a solution (the van't Hoff factor)
- C: the molar concentration of solute
- R: the ideal gas constant
- T: temperature (expressed in Kelvin)
Before exploring osmotic applications, a quick word about the van't Hoff factor. It derives from van't Hoff's law, which describes the relationship between solute concentration and osmotic pressure. This law also applies to processes of facilitated diffusion.
Examples of Osmosis
We start this section with a simple experiment you can do at home. Here's what you need to conduct it.
Place a potato wedge in each bowl. Observe how quickly the wedges deflate; the more concentrated the solute is, compared to your control wedge.
While you wait, you can rehydrate raisins (deflated grapes) by soaking them in water.
No solute needed!
After 20 minutes, one at a time, fish each wedge out of its bowl, dry it, and weigh it, starting with your control wedge. Do you note any decrease in weight in the other wedges compared to the control? Does the solute concentration have an impact on the proposed decrease?

Applications of Osmosis
Throughout this article, we've made no secret that osmosis works both in nature and in industrial applications. From water filters that hold back impurities to our kidneys, which hold back water, osmosis applications serve vital functions. These are today's most intriguing breakthroughs in forward and reverse osmosis4.
Reverse osmosis
- water desalination
- industrial water purification
- ultrapure water (pharmaceuticals and electronics production)
- food processing (juice concentration)
Forward osmosis
- wastewater treatment
- algae biofuel production
- to dilute brine solutions
- sustainable irrigation
- as a source of power generation.
It's remarkable how scientists and engineers model our most complex industrial systems on nature constructs. As these applications show, the difference between forward and reverse osmosis is impactful. As profound and impactful as the difference between active and passive transport in organisms.
Further Reading
- Osmosis. “Osmosis | Forward Deployed Reinforcement Learning Platform.” Osmosis.ai, 2025, osmosis.ai/. Accessed 21 Jan. 2026.
- University Hospitals. “Can You Drink Too Much Water?” Www.uhhospitals.org, 18 Jan. 2024, www.uhhospitals.org/blog/articles/2024/01/can-you-drink-too-much-water. Accessed 21 Jan. 2026.
- Pearson. “Osmotic Pressure Formula.” Pearson.com, 2022, www.pearson.com/channels/general-chemistry/learn/jules/ch-12-solutions/osmotic-pressure. Accessed 21 Jan. 2026.
- CATH, Tzahi, et al. “Forward Osmosis: Principles, Applications, and Recent Developments.” Journal of Membrane Science, vol. 281, no. 1-2, 15 Sept. 2006, pp. 70–87, www.statkraft.no/globalassets/old-contains-the-old-folder-structure/documents/forward-osmosis-in-jms_tcm9-24575.pdf, https://doi.org/10.1016/j.memsci.2006.05.048. Accessed 21 Jan. 2026.
Summarise with AI:
















This was very helpful and useful however the image of the animal cell, specifically in the description says it is an animal cell complete with a cell wall however animal cells don’t have a cell wall. But the rest of the article is quite useful.
Thank you so much for catching that error! You’re absolutely right – animal cells do NOT have cell walls, only cell membranes.
We’ve now updated the article to correctly identify the cell membrane as the semipermeable barrier in animal cells. We really appreciate you taking the time to point this out and help us maintain accuracy.
Thanks for reading! 😊